Steroscopic microscope
Sterioscopic Microscope SOP, K. Cayemitte, Nov 22, 2024
Phenomprox sem
PhenomProx SEM, F. Prada, Feb 20, 2024
Jaia bot sop
JAIA Bot Standard Operating Procedure, K. Cayemitte, Jan 20, 2024
Hobo ph temp loggers
HOBO pH & Temperature Loggers (MX2501), K. Cayemitte, May 16, 2025
Fire
Fluorescence Induction and Relaxation (FIRe), K. Cayemitte, Nov 26, 2024
Density kit
Density Kit Procedure, K. Cayemitte, Nov 26, 2024
Culture counter
Culture Counter, K. Cayemitte, May 14, 2025
Buoyant weight
Buoyant Weight SOP, K. Cayemitte, updated Nov 2025
Biomineral extraction workflow
Biomineral Protein Extraction Workflow, F. Prada & J. Drake, updated Dec 2025
Artificial seawater
Artificial Seawater
Protein assay workflow
Protein Assay (Bicinchoninic Acid (BCA) Assay, F. Prada, 2 March 2022
Ph meter
pH Meter Use, K. Cayemitte, 21 November 2024
Foraminifera care and measurements
Foraminifera Care and Measurements, K. Cayemitte, 27 November 2024
Sds page workflow
SDS-PAGE GEL
Glassware and plasticware cleaning
Cleaning Glassware and Plasticware, K. Cayemitte, 4 March 2025
Protein prediction from genome
BRAKER3: Protein prediction from genome
Creating doi for a github repository in zenodo
Creating DOI for a GitHub repository in Zenodo
Leica emspira 3
Leica Emspira 3
Respirometer manual
#Respirometer Manual
Last Revised: Mass Lab - Tali Mass - 20230521
[FireSting-O2 PyroScience] (https://www.pyroscience.com/en/products/all-meters/fso2-c4)
[OXROB10 PyroScience] (https://www.pyroscience.com/en/products/all-sensors/oxrob10)
Chamber stirring system
Protocol for Respirometry (oxygen flux in sealed chambers) using the PyroScience FireSting sensor, probes and software in a temperature controlled setting. This includes Apex controller for temperature manipulation and control of the respirometry water baths.
Contents
- Supplies
- Water Bath Setup
- Temperature Control
- Connecting the Battery
- FireSting Setup
- Software Setup
- Running the Program and Exporting Data
- Probe Calibration
- Stirring System Setup
- Filling and Securing Chambers
- Inserting Oxy-10 Probes
- Final Chamber Checks
- Water Changeover for Additional Runs
- Exporting Data
- Removing Chambers from Respirometer
- Take-Down and Clean Up
- Troubleshooting
- Chamber Oring Replacement
- Stir System Oring Replacement
- Oxygen meter and Sensors PyroScience
- OXROB10 Oxygen Probes
- [Pt100 Temperature Probes] (https://www.pyroscience.com/en/products/all-sensors/tdip15)
- [Pyroscience software of FSO2-C4 : Optical Oxygen & Temp Meter FireSting®-O2 (4 Channels] (https://www.pyroscience.com/en/downloads/laboratory-devices)
- Respirometer stand (with 4 screw-on legs) or underwater stirring plate

- Replacement motor - Maxon Motors Part # 459049 - 110182 A-max 26, 7W 6v DC motor + 144035 Planetary Gearhead, 26B, 53:1
- Replacement Switch
- Silicon - DuPont Molykote 112 High Performance Silicon Food Grade
- Stir system large orings - Oil-Resistant Buna-N O-Ring 3 mm Wide, 165 mm ID
- Stir system small orings - Oil-Resistant Buna-N O-Ring 3 mm Wide, 125 mm ID
- Chambers (8 can be measured at any one time with the FireSting-O2)
- MAKE SURE BOTH TOP AND BOTTOM OF THE CHAMBERS ARE LABELLED
- Magnetic stir bars x8 (35-40mm)
- Chamber screw key - ADD PHOTO HERE
- [Battery] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/images/Battery.jpg?raw=true)
- Height 2.4” Width 5.3” Depth 1.3”
- [Battery Charger] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/images/Charger.jpg?raw=true)
- Strap Wrench
- Parafilm
- Large cooler
- Lenght 38.19 x width 17.32 x height 17.72 inches
- Chiller x2
- e.g., AquaEuro 1/10 HP SKU 1457110
- Chiller Tubing
- ID 1/2” and OD 3/4”
- Chiller Pump - Hydor Centrifical Pump 300 All-Purpose Pump, 300 GPH
- Aqueon Pro 300 Submersible Aquarium Heater, 300 Watts
- Digital Thermometer Cat 150778
- Light sensor and meter if measuring photosynthesis
- 0% oxygen calibration capsules
- Minimum Data Sheet
- Make sure you choose a tank that can contain the respirometry stand (L49cm X W31cm X H32cm) and have an extra room for all coral fragments, or use two tanks, one for the respirometry stand and the other one for all coral fragments to grow in. Fill the tank with source water, enough to just fully submerge the chambers once the chambers are placed inside (~31-32 cm high).
- [Tank set up] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/images/Tank%20set%20up.jpg?raw=true)
Measure and record the temperature and salinity of the source water (where the corals are kept).
-
Fill the cooler with source water, enough to fully submerge the chambers once the stirring rack and chambers are placed inside, and set up the temperature of that water.
-
Place an aquarium pump and a heater into each bin. The pump(s) and heater(s) must be fully submerged before powering on. Plug in each piece of equipment, making sure the outlets have an appropriate voltage. Adjust the temperature of the bins so that they approximate the water temperature experienced by the coral being measured. Power on the pump and check for water flow.
-
Add a heater to the cooler The heater(s) must be fully submerged to the indicated locations for your brand of heater before powering on, or they can melt or crack
-
While you wait for the bath and source water to reach the desired temperature, set up the FireSting O2 and prepare the stirring system and chambers.
- Make sure the battery is charged.
- Unscrew the cap of the sealed [battery compartment] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/images/Battery%20compartment.jpg?raw=true) on the underside of the respirometer and locate the red and black cables. Attach the red clip to the positive terminal (marked in red) on the battery and the black clip to the negative terminal (marked in black). Test your connection and battery power by turning the switch on the other side of battery compartment [clockwise] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/images/battery%20power.jpg?raw=true). The conveyor belt should start moving at a quick pace. Turn the switch off.
- If the belt does not move or moves slowly, the battery needs to be switched out and recharged.
-
As an additional check, you can unscrew a chamber base, insert a stir bar in the center divet of the base, then reseal the chamber. Place the chamber in one of the chamber slots and note the rotation speed of the stir bar. If the stir bar is spinning slowly or not at all, the battery needs to be switched out and recharged.
- Place the connected battery inside the compartment gently and screw the lid on tightly, making sure the o-ring is properly positioned. Use a boa grip to tighten the lid with 1-2 quarter turns, then wrap with parafilm around the lid to seal the cap as a backup.
- The connection point of the wires are very delicate. Rough handing or too much tension on the wires from twisting may disconnect the wire from its base, requiring soldering to reattach the the metal foot of the wire to the metal point on the base. If this disconnectin occurs, see Troubleshooting.
-
Once the battery is placed inside, and everything is sealed, do one last check of power to make sure the motor is running (observe the black bands moving along their tracks at quick speed), then turn it off to conserve the battery until you’re ready to start your runs.
- Once you are sure the battery compartment is water tight, submerge the respirometer in the cooler’s water bath.
Below is an example of the entire set-up. This can be modified to be specific to whatever lab or space you are in, but the electronics need to be completely dry and far above/away from any potential water leak or water source.

Be very delicate with the oxygen probes. Leave the cap on the end when not in use and do not scratch the tip. The oxygen cord should never be coiled too tightly or bent, or the sensor will break.
- Insert each numbered Temp probe into the corresponding Temp port on the FireSting by aligning the red dots and pushing gently until the dots meet.
- Remove the red caps on the FireSting Oxygen ports and place all caps in the bag and in the Pyroscience suitcase.
- Remove the caps on the oxygen probes you are going to use and place each cap in its bag.
- Insert each numbered O2 probe into the corresponding O2 port on the FireSting by aligning the metal pin on the port with the opening on the probe end. Push in and turn the probe clockwise to catch the pin.
- Leave the black cap on the O2 probe until you’re ready to use the probe, then place these caps in the bag labeled “Oxygen Sensor Caps”.
- Plug the USB connector cord into your computer and the other end into the FireSting

Final Chamber Checks
Last checks before starting to record data:
- Stir bars spinning in place and at normal speed (If not, the battery needs to be switched out and recharged - see Take-Down instructions for proper procedures)
- Chambers are securely fastened on the respirometer stand
- No bubbles are present inside the chambers or on the sensors.
- The sensors are snug and secure in the chambers
- The correct probes are in the correct chambers.
- All the data is recorded on your datasheet (Each fragment is associated with the correct Cell number and sensor number).
FireSting Measurement Manual Pyro Workbench Software Setup
Perform instructions in the correct order!!
Set up
- Set-up one FireSting device with four oxygen sensors and one temperature sensor (Use the– Manual Pyro Workbench).
- Choose to measure oxygen concentration with units of- µmol/L and record every 10 sec.
- Measure water salinity.
- Calibrate the oxygen sensors with air saturated seawater. Insert an air pump into a glass with ~700ml seawater and let it bubble for 10 minutes. Transfer the water to a metabolic cell, insert a magnetic stirrer and the oxygen sensor, close the hole with plasteline, let it stable and calibrate
- • Insert the coral fragments to the chambers (one fragment into one cell) and leave one cell empty (blank- for measuring the oxygen production/consumption of the sea water). The plugs with the coral fragments should be held by the screw inside the chambers. Once the source water is at the desired temperature, submerge the chamber top and bottom and shake off or wipe away any air bubbles. Screw the plug with the coral fragment to the bottom of the chamber, while underwater. Then fill the other part of the chamber with water, insert a magnetic stir bar and close the chamber. Flip it, open the plastic lid (place the plastic screw in a bag) and let it fill with water, then, screw the chamber into the table. Make sure that they are tightly screwed and that the water level covers the chambers.
- Insert an oxygen sensor to each cell, through the hole at the top of the chamber. The sensor tip should be at least 2 cm into the chamber, and not more than a half of it should be submerged in the water. ![coral in the chamber] (
- Use a syringe to fill in with seawater or to take out all the air bubbles and seal the holes with plasteline (after you insert the oxygen sensor).
- Turn the stir system power switch to on, and check for proper stir bar placement. If any stir bars are not spinning or are out of place, try to place it using a stir bar stick or loosen the bolt and gently jiggle the chamber until the bar is reset in the center of the divot.
- When all chambers are ready, immerse the temperature sensor (Pt100) into the water tank.
- Cover the cells for 30 minutes in complete darkness.
- After 30min of acclimation to the dark, oxygen conc. will be measured first in the dark and then in six increasing light intensities (50, 100, 200, 300, 400, and 600 µmol photons/m-2/s-1), which will elevate every 15min.
Oxygen sensor installation
Be very delicate with the oxygen probes. Leave the cap on the end when not in use and do not scratch or touch their tips. The oxygen cord should never be coiled too tightly or bent, or the sensor will break.
- Remove the red caps on the FireSting Oxygen ports and place them in a bag and in the Pyroscience suitcase.
- Remove the caps on the oxygen probes you are going to use and place each cap in its bag.
- Calibrate the temperature (Pt100) and oxygen sensors [Use the Pyro Workbench & Data Inspector manual] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/Files/Manual%20Pyro%20Workbench.pdf) and measure water salinity:
- Calibrate the Pt100 temperature sensor first [use the Optical Temperature Sensors Manual] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/Files/Manual_Optical_Temperature_Sensors%20(1).pdf)
- Calibrate the oxygen sensors [use the oxygen sensors manual] (https://github.com/talimass/Talimass_Lab_Notebook_Mass_Lab/blob/master/Files/Manual_FSO2-C.pdf)
- 2-point calibration (optional): upper and 0% calibration; recommended for measurements at low O2 and high accuracy measurements over the full range.
- For seawater saturated with air, insert an air pump into a glass with 600ml seawater and let it bubble for 10 minutes. Insert the oxygen and temperature sensor into the glass and ensure that the sensor tips are immersed in the water and are free of air bubbles then measure it and let it stable. For de-oxygenated water Fill 50ml of demineralized water (DO NOT USE SEAWATER FOR THIS CALIBRATION) into a glass flask (e.g. Duran flask)
Check all orings connecting the drive motor to the stirplate for cracks. Lubricate with silicon before each run as needed
- Unscrew the cap of the sealed battery compartment on the underside of the respirometer and locate the red and black cables. ADD PHOTO HERE Attach the red clip to the positive terminal (marked in red) on the battery and the black clip to the negative terminal (marked in black).
- The connection point of the black wire is very delicate. Rough handing or too much tension on the wires from twisting may disconnect the wire from its base, requiring soldering to reattach the the metal foot of the wire to the metal point on the base. If this disconnectin occurs, see Troubleshooting.
- Test your connection and battery power by turning the switch on the other side of battery compartment clockwise. ADD PHOTO HERE The stirring system should start moving at a quick pace. Turn the switch off.
- If the system does not move or moves slowly, the battery needs to be switched out and recharged.
- As an additional check, you can unscrew a chamber base, insert a stir bar in the center divet of the base, then reseal the chamber. Place the chamber in one of the chamber slots and note the rotation speed of the stir bar. If the stir bar is spinning slowly or not at all, the battery needs to be switched out and recharged. ADD PHOTO HERE
-
Place the connected battery inside the compartment gently and screw the lid on tightly, making sure the o-ring is properly positioned. Use a boa grip to tighten the lid with 1-2 quarter turns, then wrap with parafilm around the lid to seal the cap. ADD PHOTO HERE
- Once the battery is placed inside, and everything is sealed, do one last check of power to make sure the motor is running (observe the black bands moving along their tracks at quick speed), then turn it off to conserve the battery until you’re ready to start your runs.
-
Once you are sure the battery compartment is water tight, submerge the respirometer in the cooler’s water bath and look for excessive bubbles.
- Situate the heater so it is fully submerged but not touching the respirometer stand or the Apex temp probe, and away from the battery compartment if possible.
- Consider the best placement of the temp probe, heater, and chiller outflow for adequate water monitoring and circulation. Ex. Temp probe along the back of the cooler, centered. The heater suction cupped on one far side of the cooler. The chiller inflow and outflow placed on the opposide far side of the cooler. ADD PHOTO HERE Filling and Securing Chambers

- Unscrew the bottom from a chamber and place a magnetic stir bar in the center divet. Once the source water is at the desired temperature, submerge the chamber top and bottom and shake off or wipe away any air bubbles. - ADD PHOTO HERE
- If including organisms, secure them in the bottom of the chamber before screwing the top and bottom together until hand tight. Check again for bubbles and remove any as necessary. Record the time of when you seal each chamber and leave submerged to maintain temperature until ready to secure on the respirometer. ADD PHOTO HERE
- The probes will need to reach the submerged chamber without straining the wire, insert the probes into the chamber at this time before placing the chambers onto the respirometer. - ADD PHOTO HERE
- Use the screw key to loosen the bolts of the holding plates on the respirometer just until the plates are moveable but not detached.
- ADD PHOTO HERE
- Place the chamber in a chamber slot and then insert the Oxy-10 probes.
- Be careful to not damage the probes or introduce bubbles to the chamber, insert the temperature probe into the larger probe port and gently guide it down until it fits snuggly and securely.
- Once the temperature probe is in place, gently guide the oxygen probe into its port, being careful to not scratch the probe tip. The probe tip should be at least 2 cm into the chamber, but can be inserted farther, as long as the probe isn’t touching any organisms or objects in the chamber.
- ADD PHOTO HERE
- Check for bubbles inside the chamber. If any bubbles are present, remove them at this time.
- You may be able to guide small bubbles out of the airlock seal on the chamber lid by unscrewing the airlock seal and guiding the bubbles out, then rescrewing on the airlock finger tight.
- ADD PHOTO HERE
- Note that if cooler water is not from the same source as your second bin water (used to fill the chambers), you must remove air bubbles only while submerging the chamber in the second bin’s water.
- If there are large bubbles or a large quantity of bubbles after moving the chamber, remove the chamber from the respirometer, place back into the source water, unscrew the bottom to wipe or shake away any bubbles, then close and re-secure the chamber. Note the new time of when you sealed the chamber.
- Check for bubbles inside the chamber. If any bubbles are present, remove them at this time.
- ADD PHOTO HERE
-
Repeat for all 10 chambers, filling at least one chamber with only water, without any organisms, for a “BLANK” dataset to account for background oxygen flux.
-
Once all chambers are in place, with probes and no bubbles, tighten the screws on the respirometer finger tight, making sure the holding plates are over the chamber bottoms. - ADD PHOTO HERE Test if the chambers are secure by attempting to gently move the chambers. There should be no movement.
-
Once all chambers are secure, turn the stir system power switch clockwise to on, and check for proper stir bar placement. If any stir bars are not spinning or are out of place, loosen the bolt and gently jiggle the chamber until the bar is reset in the center of the divot.
- If necessary, remove the chamber, place back into the source water, unscrew the bottom to move the stir bar into place, then close and re-secure the chamber. Note the new time of when you seal the chamber.
Last checks before starting to record data using the PreSens program:
-
Stir bars spinning in place and at normal speed (If not, the battery needs to be switched out and recharged - see Take-Down instructions for proper procedures)
-
Chambers are securely fastened on the respirometer stand
-
No bubbles are present inside the chambers
-
The correct probes are in the correct chambers (Every numbered temperature probe is with its corresponding numbered oxygen probe and in the numbered chamber you’ve assigned to the probe pair). Make sure you have recorded this on your datasheet.
-
The probes are snug and secure in the chambers
Water Changeover for Additional Runs
If using the respirometer at more than one temperature (i.e. Thermal Performance Curves)
-
Once all the chambers are out of the source water and you’ve started recording data for your current run, do a water change (if necessary) of your source water and program the Apex to your new desired temperature.
-
Once you’ve stopped recording data for your current run, program the Apex for the water bath to your new desried temperature (same as the source water).
-
While you wait for the temperature in your bath water to stabilize, collect water samples, if needed for your data set, or start preparing the chambers with the new source water using the same steps above.
Removing Chambers from Respirometer
-
Turn off the battery by turning the switch counter-clockwise.
-
Remove the probes and carefully place or drape them in a secure location.
- Unscrew the holding plates and remove the chambers.
-
YOU MUST MEASURE THE TOTAL VOLUME OF WATER IN THE CHAMBER IN ORDER TO CALCULATE THE RATE TAKING INTO ACCOUNT THE SAMPLE WATER DISPLACEMENT
-
If saving water samples, note the time when you either unscrew the airlock seal on the lid (best method for pouring the water into another container) or unscrew the bottom (keep the chamber upside down for this method).
- Unscrew the bottom of the chamber to fully drain the water, then place the two chamber parts in the source water.
- If you have any organisms secured in the bottom of the chamber, be as swift as possible to minimize time spent out of the water.
-
Keep the chamber lid very near or loosely on top of the chamber bottom, unscrewed, to not confuse which organism goes in which numbered chamber, while still allowing for water flow until you’re ready to move the chambers to the water bath.
- Once the source water and water bath are at the desired temperature, follow the same steps to fill and secure the chambers to prepare for your next run.
Respirometer Take-Down and CleanUp
- Turn off the Apex and
-
Turn off the power to the respirometer battery by turning the switch counter-clockwise.
-
Remove the probes and carefully place or drape them in a secure location.
- Unscrew the holding plates and remove the chambers.
- If collecting water samples, note the time when you either unscrew the airlock seal on the lid (best method for pouring the water into another container) or unscrew the bottom (keep the chamber upside down for this method).
-
Remove any organisms from the chambers and store or dispose of those organisms as appropriate.
-
Drain the source water bin and fill with fresh water.
- Place the unsealed chambers, stir bars, heaters, aquarium pumps, and temperature probes in the bin to soak, keeping all outlet plugs out and away from the water.
- Let the pumps run for some time (without the chiller being on) to flush fresh water through the hoses.
- Taking the respirometer out of the water bath
- When lifting the stand out of the bath, keep it tilted slightly so that the lid to the battery compartment is lower than the opposite side with the power switch.
- If any water seeped into the battery compartment, this will force the water away from other electrical components of the compartment.
- Drain the water bath and refill with fresh water. Place the respirometer in the fresh water to soak.
- While letting everything soak, collect and organize the Oxy10 probes so the Oxygen probes are together and the temperature probes are together
- Fill two beakers with DI water and gently drape the probes over a line or surface so the probe tips (any portion exposed to the water) are submerged in DI, with O2 probes in one beaker and temp probes in another.
- Breaking down the respirometer after soaking
- Lift the respirometer out of the freshwater bath using the same tilt method as before.
- Remove the parafilm and use a boa grip to loosen the cap utnil you can unscrew it by hand.
- Take out the battery and detach the positive and negative clips from the battery terminals.
- Check for any leaked water by wiping a towel, cloth, or chemwipe around the inside of the compartment.
- Place the respirometer on a stable surface to dry overnight (with the battery compartment opening-side down).
- Using a clean rag or chemwipe with ethanol, wipe down the cord of each Oxy10 probe to remove any remaining salt deposits or particulates, then recoil the probes and store in their original labeled bags.
- Be extremely careful when handling probes and make sure to avoid touching the Oxygen probe tips as they are the most sensitive.
- Leave items to soak overnight if possible, then remove from the fresh water and place on a clean surface to dry.
Disconnected battery wire
- Access the battery compartment by simply unscrewing (rotating counterclockwise) the PVC tubing which holds the battery and connections. No screwdrivers are required.
- A temporary hold may be aquired using electrical tape until you can solder the affected wire.
- A soldering iron with a fine tip is needed to reconnect the end of the wire to a small square attachment point inside the battery compartment. Before soldering, test the appropriate connection point by holding the end of the wire to the metal connection point and turning the motor ON.
- Do NOT touch any metal while the motor is on.
- Once a connection point is found, use a heated soldering iron and solder to seal that connection. Let rest for a few minutes, then test the motor by turning it ON again.
Remove bolts holding top and bottom plates together
Save all bolts and plastic spacers
Remove old orings
Replace center orings with 2 of the longer 165mm orings

Replace the outer 4 with the shorter 125mm orings

Connect to center motor hub
Replace plastic spacers
Replace top plate
Replace and tighten bolts
-
- [Putnam Lab Protocol] (https://github.com/Putnam-Lab/Lab_Management/blob/master/Lab_Resources/Equipment_Protocols/Respirometry_Protocol/Respirometry_Manual.md)
Forking the Mass Open Lab Notebook for Your GitHub
Walk-though of Forking and Modifying this Notebook Site
All steps that can be done through the terminal instead of a GUI will be written for doing in the terminal because it is good practice to use the shell
Written by Maggie Schedl
SDR microplate respirometry protocol for coral planulae
This protocol details the step-by-step use of the PreSens/Loligo Microplate respiration system for measuring respiration and photosynthesis of coral planulae.
Fixation of coral spats with resin for synchrotron radiation imaging (BAMline)
Using the EpoFix Resin and following the EMS instructions for the preparation of the resin.
Ten days old spats of the coral Stylophora pistillata were fixed with resin for synchrotron radiation imaging (BAMline) at BESSY II in Berlin.
Samples preparation
- Take one filtered tip (20 µl) and cryotube (2ml) per each sample. Cut the top part of the tip (this is where the spat will be placed) and the bottom part of the tip.
- Fit the bottom part of the tip in the lid of the cryotube. If necessary cut more of the bottom-tip to ease the interlock.
- Mix 15 parts of resin with 2 parts of hardener in a 2 ml tube. At this point bubbles will form, so use a vacuum system to remove them.
- Put a drop of resin mix at the top of the tip using a Pasteur pipette.
- Gently place the coral spat at the top of the tip using a razor blade.
- Gently add a drop of resin mix on the coral spat, wait for the resin to drip down the pipette. Add a second drop.
- After 1 hour add a third drop, make sure that the resin doesn’t drip down (the resin is almost solidified at this point).
- After 24 hours insert the tip + cup into the cryotube and close it. Your samples are ready for synchrotron radiation imaging!

Sample prep Fixation embedding and polishing protocol follow Mass et al 2017 PNAS
Standard Marine Fixation
Chemicals Needed:
10 ml ampoules 16% Formaldehyde -EMS Sciences Cat #15710 Sodium Cacodylate Trihydrate bioxtra10 g - Sigma Cat #C4945 Na2CO3 Epofix
To Make Up Solutions:
22 g/L Na2CO3 in water filtered 0.2um
0.05M Na Cacodylate Buffer in 22g/L Na2CO3 Add 10.7 gms Cacodylic acid (to 1000 ml 22g/L Na2CO3)
To Make Up Fixative:
2% Formaldehyde in Sodium Cacodylate Buffered Sea-Water Fixative Add 10 ml ampoule of 16% Formaldehyde to 70 ml of the made up buffer
Fixation Steps
- Place samples in fresh Formaldehyde fixative and keep it at RT 0.5 hrs
- Rinse with buffer (2 changes at 5 mins each)
- Dehydrate in 50,60,70, in 1g/L of Na2CO3 in 0.002M Na Cacodylate Buffer, 15 mins for each step
- 80,90, in 0.5 g/L Na2CO3 in 0.002M Na Cacodylate Buffer and 100% x2 Ethanol
- Vacuum the sample for 3 min to evaporate the ethanol.
- Usually we make up the resin (follow kit directions) when the samples are in 90% ethanol.
- Transfer into embedding resin (3 changes at 5 mins vacuum)
Embedding Steps
- Measure amount of Epofix resin needed in a Dixie paper cup. Usually 8-10 grams is enough for one sample.
- Calculate 3/25 amount of the resin to be the amount of hardener to add. Zero the balance and add the hardener into the cup. Take the cup and go upstairs.
- Stir the mixture with a glass rod vigorously and make lots of small bubbles to ensure proper mixing. It usually takes 2 minutes
- Place the cup in the desiccator (take the rod out) and pump it down for 5 minutes, then vent, and repeat 3 times. When venting, open the valve to ~2/3 of the full valve. There should be some bubbles only the surface by the end of this stage.
- Take the epoxy out of the desiccator. Use a plastic pipet to drop the epoxy into the mold with the sample in it. Avoid bubbles as much as possible. Put enough epoxy to cover the entire sample. You can use the plastic pipet to remove some bubbles if needed.
- Take the mold with epoxy filled and put it in the desiccator and repeat step 4 (pump 5 mins each for 3 times). After 3 times of pumping, leave the sample inside the desiccator for 12-14 hrs to cure.
Polishing Steps
- Start with the coarse sandpaper. Rinse with 22g/L Na2CO3 solution. Grind the back of the sample to make it flat first. Then grind the sample side until the desired surface is exposed. Add 22g/L Na2CO3 solution during the grinding to keep the sandpaper wet. Press the sample with suitable force but keep in mind that the coarse sandpaper can remove materials very fast.
- After the desired surface is approximately exposed, change to the fine sandpaper. Same as the previous step, add 22g/L Na2CO3 solution during the grinding. This step should remove all the large scratches from the last step, and the sample surface should appear foggy at the end of this step.
- After grinding, move on to polishing with aluminum oxide particles. Change the sandpaper to the felt pad and add 0.3 µm Al2O3 suspension with Na2CO3 solution. Use hand to spread it across the felt and start polishing. Add 22g/L Na2CO3 solution during the grinding. Moderate force is important: if the force is too small, it will be difficult to remove the scratches from grinding; if the force is too large, alumina particles can get stuck in the sample and create pits on the surface. Polish until a shiny mirror surface is observed and no obvious scratches can be found.
- Move on to the final polishing with 0.05 µm Al2O3 suspension. Same procedure as the previous step, but remember to change the felt to the one for fine polishing and don’t mix up with the previous pad. It’s difficult to judge if this step is done by naked eye, so you can use the microscope to help. Usually if there are no scratches under 20× it should be good enough.
- After polishing, clean the sample with a series of wipes. Prepare 4 wipes with the following order: wet with Na2CO3 solution, dry, wet with ethanol, dry. After polishing with 0.05 µm Al2O3, clean the sample through these wipes as the order above. Gently wipe once on each wipe. If there are watermarks or weird patterns on the surface, it means that the cleaning was not done properly so you need to repeat this step.
Coverslips coating for Coral tissue culture
Laminin Solution
Laminin (Sigma # L 2020, 1mg/ml) Aliquot into 20μl units Store at -20ºC • Add diluted Laminin solution to cell suspension at a ratio of 1 part Laminin solution to 3 parts cell suspension.
Sodium borate solution (0.19M)
7.6 g Na2B4O7 (Sigma # 221732) 200ml DDW pH 8.2
Poly- D- lysine solution (1mg/ml)
10mg Poly-D -lysine (Sigma# P 1024) 10ml Sodium borate solution Filter Sodium borate solution only Keep for 30 minutes at room temperature, Store at -4º
Borate Buffer pH 8.5
2.375g Na2B4O7*10 H2O (Sodium Tetraborate) (Sigma # S 9640)
1.550g H3BO3 (Boric Acid) (Sigma # B 6768)
500ml DDW
pH-8.5 with 1M HCL
Filter and store at 4◦C
PolyEthylenImine (PEI) solution (100μg/ml)
0.1ml or 0.107g 50% PEI (Sigma# P 3143, 50ml)
49.9ml Borate Buffer
Aliquot into 1ml amounts,
Store at -70◦C
Before using : thaw 1ml aliquots and add 9ml Borate Buffer
Store rest at -20◦ C for no more than 1 mounth.
Coating coverslips
Use about 50μl/cm2 Poly-D-lysine / Poly-Ethylene-Immine solution
Coat overnight at room temperature,
Aspirate solution using sterile Pasteur pipette
Wash in DDW,
Dry with sterile Pasteur pipettes.
Coral tissue culture protocol follow Mass et al 2012 PlusOne
Using the DMEM no glucose and antibiotic-antimycotics
Prepare CaFSW
Add the follow to 1l of deionized H2O and filter with 0.2µm keep stock in 4C and worm to 25C before using
| chemical comment | gram |
|---|---|
| NaCl | 23g |
| KCL | 0.763g |
| MgSO4-7H2O | 1.89g |
| MgCl2-6H2O | 10.45g |
| Na2SO4 | 3g |
| NaHCO3 | 0.25g |
| SrCl2 | 0.026g |
DMEM culture medium
Add to [DMEM no glucose] (https://www.thermofisher.com/il/en/home/technical-resources/media-formulation.49.html) the follow
| chemical comment | gram | comment |
|---|---|---|
| NaCl | 9.05g | |
| KCL | 0.7g | |
| CaCl2 -2H2O | 0.71g | |
| MgCl2-6H2O | 5.1g | |
| Na2SO4 | 0.5g | |
| taurine | 0.5m | from stock 1.04µg/20ml DDW |
| NaOH3- | 1.85g | only if necessary, if the DMEM bought is without NaOH3 |
| Hepes | 2.98g |
Keep stock in -20C
Working solution
Dilute the stock DMEM culture medium to 12.5%(vol/vol) in ASW: 20ml DMEM + 140ml ASW
Add to the diluted DMEM mix :
- 1.25% FCS
- 1% antibiotic-antimycotics (1.6ml)
- 5mM glucose (For DMEM without glucose)
- 20 µg/ml aspartic acid (0.0032g)
- 50 µg/ml ascorbic acid (0.008g)
- 1:100 L-Glutamine (1.6ml)
- Final pH 8 – monitor gently with NaOH 1M or NaCl 1M
- Filter all the medium in 0.2µm
Tissue culture preperation
All work must be done in the biological/laminar flow hood with filtered medium All tips should be stored in the hood. Tips, pipettes, etc. should be exposed to UV light for at least 10 minutes
All medium need to be in 26C
Work with 6wells or 5ml petri-dish
- Wash 0.2-0.7 cm long nubbins or fragments of nubbins for 15 min in PVP-Iodine
- 2 min wash in FASW and 1 min in DDW.
- Repeat this step if necessary (this steps are done in order to get rid of all the ciliates in medium)
- Pre Incubate nubbins for 4.5 h in CaFSW +3% antibiotics (slow rpm in room temp)
- Transfer the coral nubbin to culture medium (26C)
- Incubate in humidified chamber/incubator with 12/12 h light dark cycle at 26C for 48h.
- The tissue might fall of the skeleton and if not take it off by gentle pipetation. Take out the skeleton homogenize solution and add a drop of PVP-Iodine for 15 min.
- Transfer the medium to 15ml tube and spin down the medium contains the cells (5 min 500g in RT).
- Change the medium with fresh one
- Filter (0.2µm) the medium with the cells to a sterile plate (1ml to each plate) and add 4ml of fresh medium
- Incubate in humidified chamber with 12/12 h light dark cycle at 26C and change medium every 3 days.

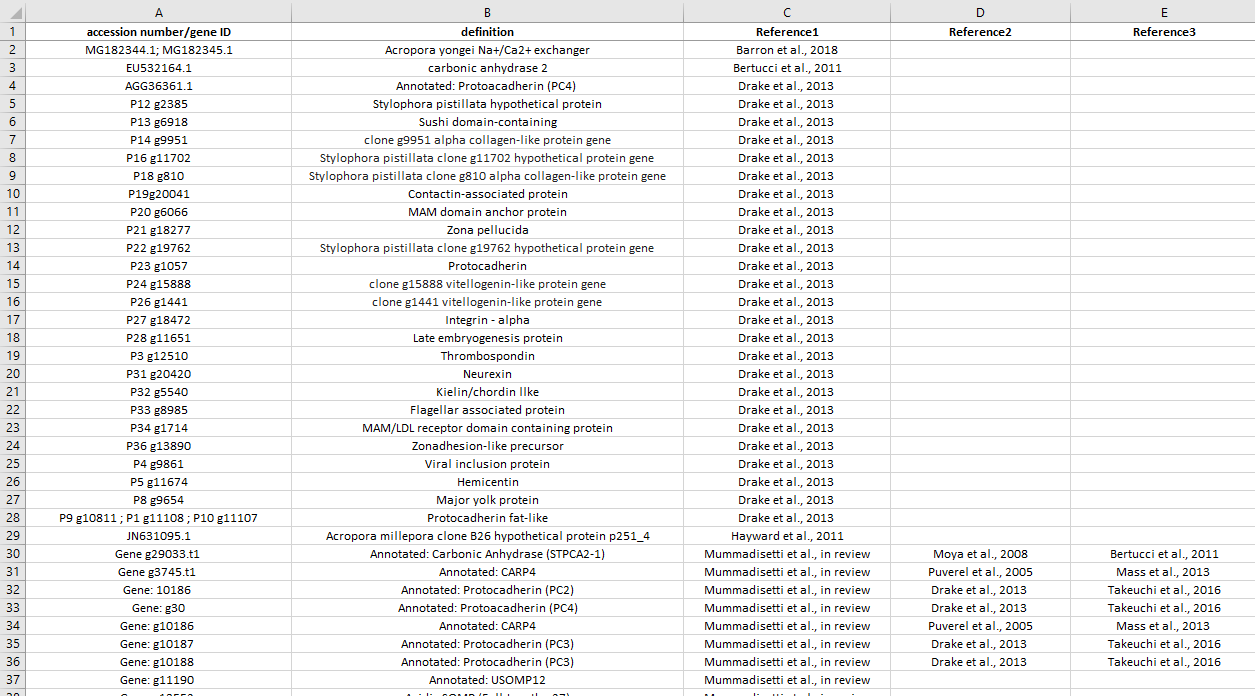
Known biomineralization-related genes list
This list comprehends all biomineralziation-related genes known from literature. The first reference is the one related to the accession number/gene ID, then we put all other references where that specific gene/protein was previously detected/immunolocalized (chronological order).
List preview:
Protocol for coral physiology
These instructions allow to quantitatively assess coral physiological parameters, i.e. tissue biomass (protein concentration), algal density and chlorophyll concentration.
Count coral symbiont cells with Fiji (ImageJ)
These instructions cover two ways to get algal cell counts using Fiji, one manual and one automated. A fluorescence microscope (Nikon Eclipse Ti, Japan) was used to image symbiotic algae (isolated from coral tissue) both in brightfield and in fluorescent light using 440 nm emission, to identify chlorophyll and to ensure counting of symbiont cells only.


